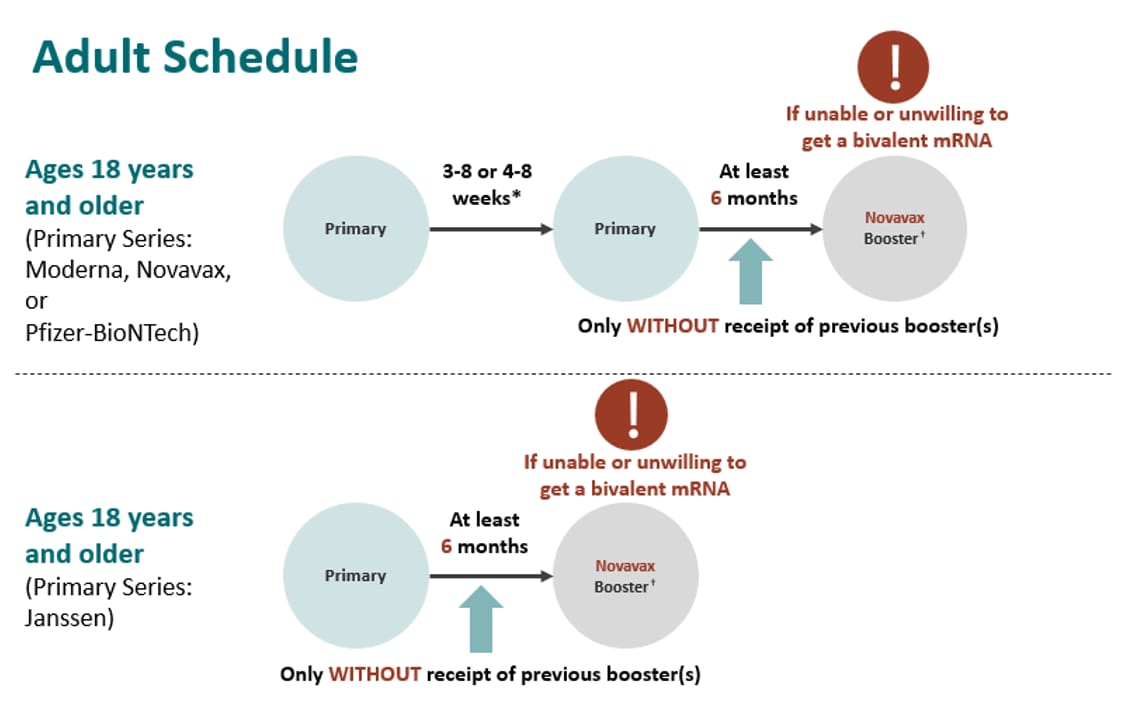
Novavax on X: "NEWS: Novavax Announces #COVID19 Vaccine Booster Data Demonstrating Four-Fold Increase in Neutralizing Antibody Levels Versus Peak Responses After Primary Vaccination https://t.co/hHkag8Nm6b https://t.co/zlN24CliAR" / X

Novavax Expects Its Updated COVID-19 Vaccine Candidate to Work on Circulating Variants | Health News | U.S. News